39 eu language requirements for product labels
Product Labelling — EUbusiness.com | EU news, business and politics Almost all products sold within the EU will require some form of labeling, but what details are required depends on the nature of the product. Here are a few examples of the topics covered by EU legislation: EU Food Product Labeling; General legislation covering labeling, presentation and advertising Nutritional Alcoholic beverages EU Labeling Requirements - United States Mission to the European Union Nutrition labeling becomes mandatory on December 13, 2016 Minimum font size for printing mandatory information New format for allergen labeling (allergens must be highlighted in the list of ingredients - "allergen boxes" are no longer allowed) Voluntary front-of-pack labeling has to follow a set format
What is the legislation in Europe for the languages to use on labels ... According to EU food (and water and beverage) legislation food labels must be given in a language that is understandable by consumers in the country in which you are selling your products. Therefore English will be good in the UK (and in Malta!) but not throughout Europe. This is the situation in EU countries but also the rest of the world.

Eu language requirements for product labels
Country Language 1 Language 2 Language 3 - Europa 1)Please check with the local authorities, requirements can vary depending on region. 2) German OR French 3)The labelling must be written in at least two official languages. With the agreement of individual professional final users, a substance for supply to these final users may be labelled in only one official language or in English. Official Language in EU required for Medical Device Labeling User interface Language Requirements - EU MDR: EU Medical Device Regulations: 1: Nov 6, 2020: C: Non-EU Language Requirements: Other Medical Device Regulations World-Wide: 3: Aug 10, 2020: K: China Medical Device Labeling requirements - Language: China Medical Device Regulations: 9: Jun 15, 2020: The unbearable insensitivity of risk management ... European Union - Labeling/Marking Requirements (part 1) EU - export Directive 2010/30/EU "on the indication by labeling and standard product information of the consumption of energy and other resources by energy-related products" sets a framework for the adoption of product-specific directives on the proper energy efficiency labeling for each concerned product.
Eu language requirements for product labels. Labels and markings - Your Europe Most footwear sold in the EU must bear a label that informs potential buyers what they are made of. Footwear Label Clothes and other textile products sold in the EU are required to carry a label with information on the textile fibre composition. This enables your customers to make an informed decision when they buy. Textile Label Switzerland - Labeling and Marking Requirements Generally, labeling and marking requirements follow EU regulations (CE labeling); however, a CE mark is not required for a product made only for Swiss domestic use. SECO coordinates the implementation of the Federal Act on Product Safety and more information can be found on their webpage on product safety (available in German, French, and Italian). Product-information requirements | European Medicines Agency EMA's guidance explains the content that should be included in these documents, as well as standard headings and the most commonly used standard statements and terms in all official European Union (EU) languages plus Icelandic and Norwegian, and defines the format and layout for the product information. EMA's guidance is without prejudice to: Access2Markets Labelling and packaging - Europa The information provided by labels must be easy to understand, easily visible, clearly legible and indelible and must appear in the official language (s) of the Member State where the product is marketed. However, the use of foreign terms or expressions easily understood by the purchaser may be allowed. List of applicable legislation
How to Create a Label as per EU MDR 2017/745? Innovations by MDR 2017/745. Manufacturers of medical devices are subject to a number of new requirements as a result of the EU Medical Device Directive, mainly the following: The scope of application also extends to non-medical products (e.g. contact lenses, devices for liposuction). Each medical device must bear a unique identification number ... European language labeling for Medical Devices CE Mark EU Foreign Language Labeling Requirements. There are two major areas of confusion about the translation requirements when CE marking a product for export to the EU States. One is that everyone talks about "CE Marking translation requirements" without actually reading the specific Directive(s) that apply to their products. Product Labeling Requirements: What You Need To Know The US, Canada, Mexico, and the EU all require that your product packaging be written in local languages. In Canada, that means your labels need to be in English and French. In Mexico, that means Spanish. Choosing a Labeling Translation and Compliance Partner Textile Label - Your Europe Every textile product must be labelled or marked to show its fibre composition whenever the product is marketed in the EU. These labels must be firmly attached to the product, for example, sewn in. This requirement concerns all products made up of at least 80% of textile fibres, calculated by weight, such as: clothing furniture coverings
Product Labeling Regulations in the US, EU and Australia Here are few labeling requirements for imported products: a. The label must contain the identity of the prepackaged product in terms of its function or generic name. b. The label must contain the principal place of business and identity of the manufacturer or dealer. c. European Language Translation Requirements for Medical Device Labeling ... An * indicates that that English may be acceptable for devices used only by healthcare professionals. Even so it is always recommended to translate into an official language of that country. AUSTRIA - German BELGIUM - Dutch, French and German BULGARIA - Bulgarian* CROATIA - Croatian CYPRUS - Greek* CZECH REPUBLIC - Czech DENMARK - Danish EU product requirements | European Commission Requirements for the use of chemicals (REACH) How to comply with the EU's classification, labelling and packaging regulation, how to classify chemicals, submitting a substance to the classification and labelling inventory. Classification, labelling, packaging of chemicals Details of chemical products that require additional or specific legislation. EU - Labelling Requirements | CE Intelligence The use of language on labels has been the subject of a Commission Communication, which points out that labelling of foodstuffs for sale to the final consumer must be in an easily understandable language which is generally interpreted to mean the language of the country of marketing (European Commission ,2010).
EU MDR language requirements — what manufacturers and ... - Decomplix In the explanations on technical documentation in Annex II, there is also a list of the information that the manufacturer must include in the respective official languages, namely the label (s) on the product and its packaging (single unit packaging, sales packaging, transport packaging) as well as the instructions for use. GUI for medical software
EU Cosmetic Labeling Requirements | CosmeReg What are the requirements for EU cosmetic labeling? To successfully place your cosmetic product on the EU, read our article! Kakaotalk ID: Cosmereg +44 20 33182439 +1 727 3509380; ... You must translate the function of the product into the native language of each country the product is being introduced to.
United States Product Labeling Requirements: An Overview - Compliance Gate The Federal Trade Commission (FTC) establishes labeling requirements for importers and manufacturers to adhere to with regard to clothing and textile products. For example, labels on clothing and textiles should be obvious, accessible, and contain information such as the following: Fiber content (e.g. 100% cotton)
EU - Labeling/Marking Requirements - International Trade Administration Effective July 16, 2021, the EU required all CE marked products to have a label that identified a point of contact within the region. This requirement applies to products sold online and through traditional distribution channels.
EU Language Requirements | Obelis EU Language Requirements EU Language Requirements The label of a cosmetic product (container & outer packaging) should follow the language requirements which are exerted by the National Laws of the EU member states.
Labelling and packaging - ECHA - Europa Labelling and packaging. Once the hazardous properties of a substance or mixture have been identified, they need to be classified accordingly. Manufacturers, importers, downstream users and distributors, as well as producers and importers of certain specific articles, must communicate the identified hazards to the other actors in the supply ...
As packagers consider these and other EU MDR A medical device labelling product compliance requires tedious paperwork and detailing, right from the application process until the final approval of the label. Manufacturers/importers often find it difficult to keep track of all the requirements. At CliniExperts, we ensure that your medical device labels comply with the. Option 1: Distribution.
European Union Product Labelling Requirements: A Complete Guide EU Textiles Labelling Clothing and other products containing a minimum of 80% by weight of textile fibres must be labelled with the correct fibre composition (e.g. 100% Cotton or 100% Polyester). Further, the label must be permanent, which means it must either be attached to the clothing item or printed. A sticker is not enough. Product Examples
EU: Language Requirements for Product Labels - GlobalTrade.net Belgium has three official languages: Dutch, French and German. All information which is required to appear on labels must be in the official language of the region where the product is commercialized (if necessary, further requirements for specific products may be obtained from the importer).
EU labels | European Commission EU Ecolabel- Products covered by the EU's Ecolabel initiative, criteria for establishing an Ecolabel, how to apply for an Ecolabel, application and annual fee rates. Energy labels. Energy efficient products - Requirements for energy efficient products, EU energy labelling and ecodesign rules, the EU's energy star programme.
European Union - Labeling/Marking Requirements (part 1) EU - export Directive 2010/30/EU "on the indication by labeling and standard product information of the consumption of energy and other resources by energy-related products" sets a framework for the adoption of product-specific directives on the proper energy efficiency labeling for each concerned product.
Official Language in EU required for Medical Device Labeling User interface Language Requirements - EU MDR: EU Medical Device Regulations: 1: Nov 6, 2020: C: Non-EU Language Requirements: Other Medical Device Regulations World-Wide: 3: Aug 10, 2020: K: China Medical Device Labeling requirements - Language: China Medical Device Regulations: 9: Jun 15, 2020: The unbearable insensitivity of risk management ...
Country Language 1 Language 2 Language 3 - Europa 1)Please check with the local authorities, requirements can vary depending on region. 2) German OR French 3)The labelling must be written in at least two official languages. With the agreement of individual professional final users, a substance for supply to these final users may be labelled in only one official language or in English.




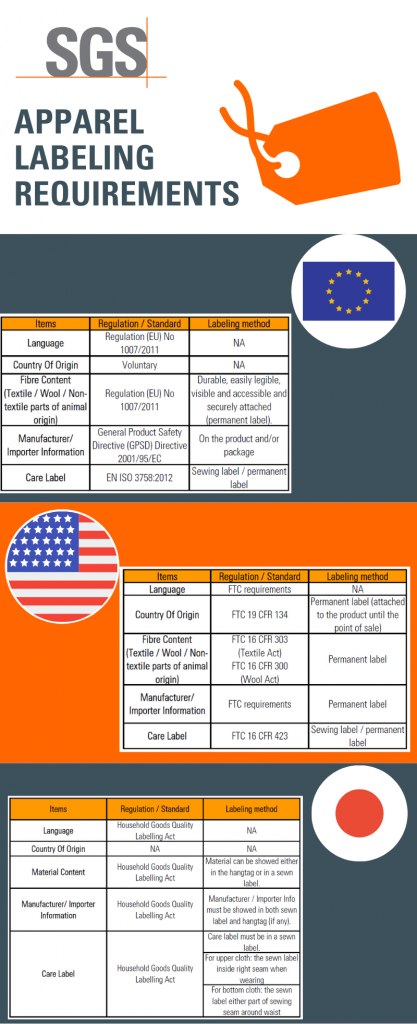
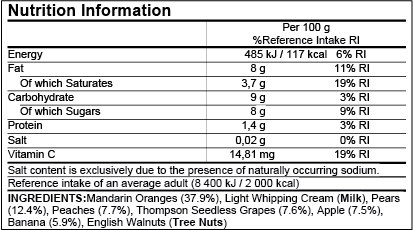
















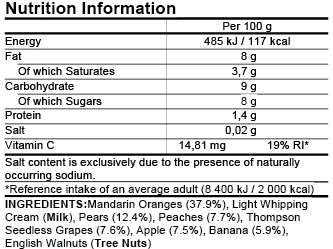





Post a Comment for "39 eu language requirements for product labels"